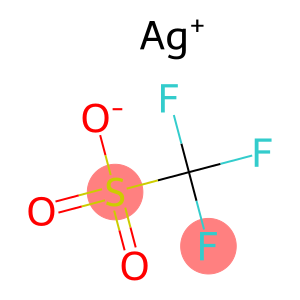
SILVER TRIFLATE
silver trifluoromethanesulfonate
CAS: 2923-28-6
Molecular Formula: CAgF3O3S
SILVER TRIFLATE - Names and Identifiers
| Name | silver trifluoromethanesulfonate |
| Synonyms | SILVER TRIFLATE Silver triflate SILVER TRIFLUOROMETANESULFONATE Silver trifluormethanesulfonate silver trifluoromethanesulfonate SILVER TRIFLUOROMETHANESULFONATE SILVER TRIFLUOROMETHANESULPHONATE SILVER(I) TRIFLUOROMETHANESULPHONATE silver(1+) trifluoromethanesulfonate SILVER (I) TRIFLUOROMETHANE SULFONATE TRIFLUOROMETHANESULFONATE SILVER SALT TRIFLUOROMETHANESULFONIC ACID SILVER SALT Trifluoromethanesulfonic acid, silver salt Silver(I) triflate~Trifluoromethanesulphonic acid silver(I) salt |
| CAS | 2923-28-6 |
| EINECS | 220-882-2 |
| InChI | InChI=1/CHF3O3S.Ag/c2-1(3,4)8(5,6)7;/h(H,5,6,7);/q;+1/p-1 |
| InChIKey | QRUBYZBWAOOHSV-UHFFFAOYSA-M |
SILVER TRIFLATE - Physico-chemical Properties
| Molecular Formula | CAgF3O3S |
| Molar Mass | 256.94 |
| Melting Point | 286 °C (lit.) |
| Boling Point | 162°C at 760 mmHg |
| Water Solubility | Soluble in water and also in most organic solvents. |
| Solubility | Methanol (Slightly), Water (Slightly) |
| Vapor Presure | 1.14mmHg at 25°C |
| Appearance | Bright light brown crystal |
| Color | White to light beige |
| BRN | 3598402 |
| Storage Condition | Store below +30°C. |
| Stability | Stable, but may be light sensitive. Incompatible with strong acids, strong oxidizing agents. |
| Sensitive | Light Sensitive |
| MDL | MFCD00013226 |
SILVER TRIFLATE - Risk and Safety
| Risk Codes | R36/37/38 - Irritating to eyes, respiratory system and skin. R50 - Very Toxic to aquatic organisms R34 - Causes burns |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36 - Wear suitable protective clothing. S61 - Avoid release to the environment. Refer to special instructions / safety data sheets. S45 - In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.) S36/37/39 - Wear suitable protective clothing, gloves and eye/face protection. |
| UN IDs | 3260 |
| WGK Germany | 3 |
| FLUKA BRAND F CODES | 8 |
| TSCA | Yes |
| HS Code | 28432900 |
| Hazard Note | Corrosive |
| Hazard Class | 8 |
SILVER TRIFLATE - Introduction
Soluble in water.
Last Update:2022-10-16 17:24:19
SILVER TRIFLATE - Reference Information
| EPA chemical information | Information provided by: ofmpub.epa.gov (external link) |
| Use | Reagents for halogenation with trifluoromethanesulfonic acid; glycosylation reagent of halogenated glycosyl; deprotection with thiol as protective group Reagents; The reaction of producing olefins with hypophosphate and thiophosphate; Reagents for etherification of halogenated hydrocarbons and alcohols under mild reaction conditions; Used to generate ion catalyst from rhodium chloride complex rhodium; applied to the acetylene of hydrogenated phosphorous acid; a catalyst for the preparation of silyl ether by the hydrosilylation of aldehydes |
| production method | should react in the dark. In a 500ml bottle, 300ml of water is filled, 60g(0.4mol) of trifluoromethanesulfonic acid is slowly added, and the bottle is filled with a magnetic stirrer coated with polytetrafluoroethylene. Put the bottle in an ice bath to cool. 48.5g(0.21mol) of powdered silver oxide was added in installments. Remove the cold bath, continue stirring in the dark for 25min, and filter out the suspended matter from the porous glass sand core funnel. The filter residue is washed with 100ml distilled water, and the residue is dissolved in 250ml of acetone. The acetone solution was filtered through a porous glass sand core funnel. The solvent was evaporated, the product was ground into fine powder, and dried at 100 ℃ for 6 hours at 3.32Pa to obtain 87.9g(98%) of silver trifluoromethanesulfonate. The product should be stored in a dryer to avoid light. Silver trifluoromethanesulfonate can also be made from the reaction of barium trifluoromethanesulfonate with silver carbonate. |
Last Update:2024-04-10 22:29:15
Supplier List
Featured ProductsMultiple SpecificationsSpot supply
Product Name: Silver trifluoromethanesulfonate Request for quotationCAS: 2923-28-6
Tel: 15001012717
Email: biz5@infsci.com
Mobile: 15001012717
QQ: 3007471201
Product List: View Catalog
Spot supply
Product Name: Silver trifluoromethanesulfonate Visit Supplier Webpage Request for quotationCAS: 2923-28-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: silver trifluoromethanesulfonate Request for quotation
CAS: 2923-28-6
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 2923-28-6
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Spot supply
Product Name: Silver trifluoromethanesulphonate Visit Supplier Webpage Request for quotationCAS: 2923-28-6
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Spot supply
Product Name: silver trifluoromethanesulfonate Request for quotationCAS: 2923-28-6
Tel: 86+027-83389957
Email: 1024042217@qq.com
Mobile: 86+18627766980
QQ: 1024042217
Wechat: 18627766980
Product Name: SILVER TRIFLUOROMETHANESULFONATE Request for quotation
CAS: 2923-28-6
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 2923-28-6
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Spot supply
Product Name: Silver trifluoromethanesulfonate Visit Supplier Webpage Request for quotationCAS: 2923-28-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Featured ProductsMultiple SpecificationsSpot supply
Product Name: Silver trifluoromethanesulfonate Request for quotationCAS: 2923-28-6
Tel: 15001012717
Email: biz5@infsci.com
Mobile: 15001012717
QQ: 3007471201
Product List: View Catalog
Spot supply
Product Name: Silver trifluoromethanesulfonate Visit Supplier Webpage Request for quotationCAS: 2923-28-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: silver trifluoromethanesulfonate Request for quotation
CAS: 2923-28-6
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 2923-28-6
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Spot supply
Product Name: Silver trifluoromethanesulphonate Visit Supplier Webpage Request for quotationCAS: 2923-28-6
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Spot supply
Product Name: silver trifluoromethanesulfonate Request for quotationCAS: 2923-28-6
Tel: 86+027-83389957
Email: 1024042217@qq.com
Mobile: 86+18627766980
QQ: 1024042217
Wechat: 18627766980
Product Name: SILVER TRIFLUOROMETHANESULFONATE Request for quotation
CAS: 2923-28-6
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 2923-28-6
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Spot supply
Product Name: Silver trifluoromethanesulfonate Visit Supplier Webpage Request for quotationCAS: 2923-28-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History






